Press Release: ScitoVation to Showcase ScitoSim™ AI Platform at Society of Toxicology (SoT) 2026
Read More!
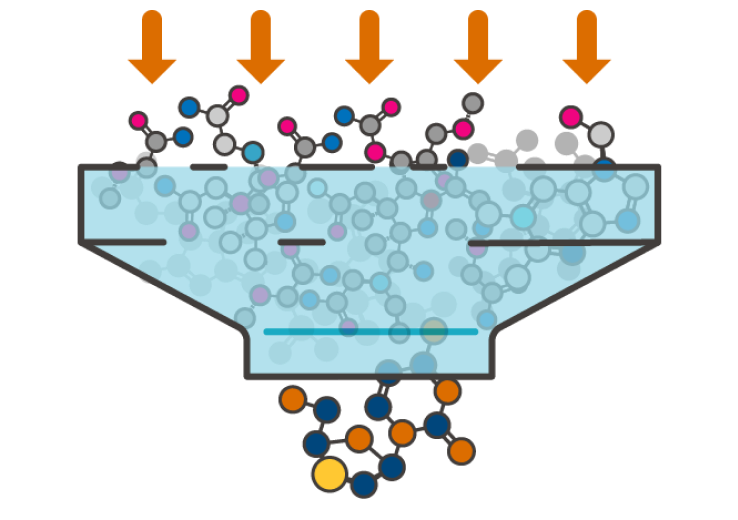
A CRO, ScitoVation helps clients assess the safety and efficacy of pharmaceuticals, chemicals, food ingredients, cosmetics, and other consumer products faster using new approach methods (NAMs)
What our Customers are saying
I’ve been closely engaged with ScitoVation on projects for the last 3 years, helping a consortium of 14 global companies address a generic problem that impacts all of them. These companies either don’t have the expertise or can’t dedicate a resource to address the challenge. ScitoVation’s creative approach using PBPK modeling is notable because not only are they innovative, but they are also accountable with budgets and timelines, practical and easy to work with.
How We Serve Our Clients
Meeting the Needs of Clients
Our team of pioneers is known for taking the time to understand the problem of each client and create an approach that best addresses their challenge. In many cases, we create custom approaches to help the client move quickly to their next research stage. This could be a go or no go decision, mode of action, or submission to a regulatory body. Our clients not only benefit from our expertise, but also time and cost savings.